

Faded Silver
Faded Silver
When a film is duplicated for preservation the image lost to fade cannot be recovered. Mick Newnham investigates a process to intensify the faded silver image to enable the duplicated image to be nearer to the original in density and contrast without exposing the emulsion to the risk of chemical treatments.
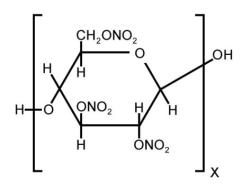
From the earliest days of motion pictures until the early 1950s, cellulose nitrate was the dominant polymer used for professional motion picture film base. In the presence of moisture cellulose nitrate decomposes via a hydrolytic pathway and releases a range of oxides of nitrogen, some of which combine with moisture to form nitrous and nitric acids.
The first stages of decomposition include the fading or yellowing of the silver image due to oxidation by the nitrogen oxides to colourless silver compounds.
Traditional techniques for intensifying a silver image involve aqueous solutions. However the gelatin emulsion of a decomposing film is not sufficiently stable to withstand the treatment and will generally disintegrate if it is exposed to aqueous solutions. By vacuum pre-treatment and exposing to specific wavelengths of light the silver compounds can be reduced to metallic silver to substantially reform the original image.
Nitrate decomposition
Cellulose nitrate is a cellulose ester manufactured by substituting nitro groups for the hydroxyl groups on the glucose ring (Figure 1 above). There are many different uses for cellulose nitrate. Some cellulose nitrates have different degrees of substitution to achieve the characteristics best suited to a specific purpose (Miles, 1955; Reilly, 1991).
In the presence of moisture, cellulose nitrate used in motion picture film base decomposes via an acidic hydrolysis pathway and releases a range of oxides of nitrogen (Carroll, 1955), some of which combine with available moisture to form nitrous and nitric acids. The first stages of decomposition include the fading or yellowing of the silver image due to oxidation by the nitrogen oxides to colourless silver compounds.
Film archivists describe the decomposition of nitrate film as occurring in five stages (FIAF, 1992):

- D1 - image fade;
- D2 - severe image fade and stickiness;
- D3 - honey, a partially digested byproduct from the nitric acid's action on cellulose;
- D4 - blocking, the internal adhesion of the film pack; and
- D5 - red dust, cellulose powder and colloidal silver.
During the first two stages, D1 and D2, the film is still sufficiently stable to be handled and possibly duplicated.
The characteristic image fade of the D1 stage is via a process of silver oxidation, initiated by the acidic environment.
Understanding silver
Silver is the basis of traditional photography and understanding silver deterioration is crucial to understanding the preservation of black-and-white film. In the typical filamentary structure of developed photographic silver there are a large number of reactive sites where oxidation and other reactions can occur (Brandt, 1987).
The common reactions between silver and decomposition byproducts are (Carroll, 1955):
- Nitrogen dioxide reacts with silver to form silver nitrate: 2NO2 + Ag -> NO + AgNO3
- Nitrogen dioxide also reacts with water to form nitrous and nitric acid: 2NO2 + H2O -> HNO3 + HNO2
- Nitric acid reacts with silver to form silver nitrate and water: 2HNO3 + 2Ag -> AgNO3 + AgNO2 + H2O
The action of nitric acid on silver is accelerated by the presence of nitrous acid (Carroll, 1955).
The physical characteristics of a roll of film within an enclosure, usually a film can, lead to the formed nitric acid being held within the film pack with a low potential for diffusion (Newnham, 2002 and 2003). This is in contrast to the diffusion potential from a large-format sheet of nitrate film. The increased surface area-to-volume ratio of sheet film creates a greater potential for diffusion of the decomposition products from the base.
The nitric acid environment maintains the predominant silver compound as silver nitrate:
Ag2O + 2HNO3 -> 2AgNO3 + H2O
'The order in which the film components are attacked by nitrogen dioxide is: the silver or dye first, the gelatin of the emulsion second’ (Carroll, 1955). The gelatin emulsion is pH sensitive; as the pH drops the gelatin viscosity decreases and the solubility increases significantly (Sheppard, 1927; Sheppard and Houck, 1930). In practice, the stability of the emulsion is so compromised by the action of the nitric acid that any attempt to use a traditional aqueous developer solution to reduce the silver compounds would end in the complete destruction of the emulsion and consequently the image.
Enter light
Silver nitrate is a light-sensitive salt. While the film remains in a wound reel, light cannot enter the film pack to cause the photo-reduction of the silver nitrate. To reduce the silver nitrate to metallic image-forming silver, an exposure to an ultraviolet/blue-rich light source is required.
Cellulose chains will undergo scission in the presence of silver nitrate when exposed to very short wavelengths, around 250 nm (Allen, 1995). To avoid further degradation of the film base and the issues surrounding working with ionising radiation, longer wavelengths that still have sufficient actinic energy to reduce the silver are required.
A wavelength of around 350 nm was chosen as the centre of a suitable range: first, as a result of the work by United States Army Research Laboratory in the formation of silver clusters (Gaddy, 2004); and second, because inexpensive and readily available light sources rich in this spectral region could be purchased.
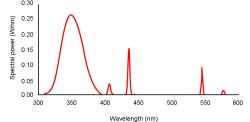
Fluorescent tubes with a peak output around 350 nm are used in electronic insect traps. A typical output curve of these types of light sources is shown in Figure 2.
The spectral power of these tubes is quite low. However in the experimental system there was sufficient energy to reduce the silver nitrate to silver metal because:
- of the close proximity of the tubes reducing the effects of the inverse square law; and
- silver nitrate is more readily reduced in the presence of organic materials, in this instance the gelatin emulsion.
The reduction of silver nitrate is a readily reversible reaction, especially in an environment with a high concentration of nitric acid. To minimise the reformation of silver nitrate the excess nitric acid was removed using a vacuum treatment. The film was placed in a vacuum desiccator connected to a vacuum pump. The film was held at reduced pressure for approximately 24 hours.
This treatment had an additional effect of removing some of the oxygen and water from the film emulsion, both active in silver oxidation. The vacuum treatment was an important step in the process and is analogous to hypersensitising used by astronomers to increase the effective film speed of film emulsions without resorting to higher-speed film with a coarser grain structure (Wilson, 2001).
Image intensification
The intensified silver image is formed by colloidal silver (Gavrilyuk and Lanskaya, 2006) and consequently has a warm, reddish appearance. The image is slow to form and determining a suitable end point to the reduction reaction is problematic. While the silver reduction is complete when all of the reducible silver salt has been reduced, there remains the highly probable risk of damage to the nitro cellulose ester film base if the exposure is extended and the risk, while less likely, of other Maillard-like reactions within the deteriorated gelatin emulsion that may cause discolouration and an overall density increase.
The results are both satisfying and disappointing. While the silver compounds can be successfully reduced in situ without traditional developers, the image is insufficiently sharp due to migration within the emulsion of the silver ions through various stages of oxidation over the life of the film (Reilly, 1988).


Under close examination the halo artefact due to migration of the oxidised silver through the emulsion can be clearly seen in Figure 4. While the effect of the intensification is sufficiently stable to permit duplication, the colloidal silver is readily oxidised. Within a few weeks of intensification, oxidation 'Red spot’ (Reilly, 1988) blemishes were apparent in several areas of the image. Red spot blemishes can also be seen in Figure 4
Conclusion
While the treatment is fairly straightforward it is slow and time consuming and, due to the diffusion of silver ions over time, the resultant image is less than sharp. However nitrate motion picture film contains many unique records. Some of these records have a very high value in terms of historical or other measures. This treatment may help recover lost information or enable a faded image to be more easily interpreted.
References
Allen N (1995) Verification of the Nature and Causes of the Photo-negative Images on the Shroud of Lirey-Chambery-Turin, University of South Africa.
Brandt E (1987) 'Mechanistic Studies of Image Stability. 3. Oxidation of Silver from the Vantage Point of Corrosion Theory’, Journal of Imaging Science, vol. 3, no. 6.
Carroll J and Calhoun J (1955) 'Effect of Nitrogen Oxide Gases on Processed Acetate Film’, Journal of Society of Motion Picture and Television Engineers, vol. 64, no. 9, pp 501-507.
FIAF (1992), Handling, Storage and Transport of the Cellulose Nitrate Film, International Federation of Film Archives, Brussels.
Gaddy G (2004) Reversible Formation of Silver Clusters and Particles in Polymer Films, US Army Research Laboratory.
Gavrilyuk AI and Lanskaya TG (2006) 'Photochromism in RbAg4I5 Thin Films Enhanced by Hydrogen Photosensitisation Simultaneous to Illumination’, Thin Solid Films, vol. 515, issue 4, pp 2337-2341.
Ligterink F (1999), Application of The Hoetjer Equation in Climate Control Studies, IAP 1999, Indoor Air Pollutants Working Group, Netherlands.
Miles F (1955) Cellulose Nitrate, Oliver and Boyd, London.
Newnham M (2002) 'Ventilated Film Cans – Their Effect on the Diffusion of Decomposition By-products from Motion Picture Film’, Journal of the Society of Motion Picture and Television Engineers, vol. 111, no. 1, pp 29-33. A version of this paper is viewable at NFSA Preservation and technical research.
Newnham M (2003) 'Investigation into the Mechanism of Maintaining a Lower Free Acid Level Inside CTA Motion Picture Films During Long Term Storage Using a Low Tension “Preservation” Wind’, Journal of Film Preservation, vol. 66 (October), pp 37-44.
Reilly JA (1991) 'Celluloid Objects: Their Chemistry and Preservation’, Journal of American Institute for Conservation, vol. 30, no. 2.
Reilly JM (1988) 'Stability of Black and White Photographic Images, with Special Reference to Microfilm’, Abbey Newsletter, vol. 12, no. 5.
Sheppard S (1927) 'Behaviour of Gelatin in the Processing of Motion Picture Film’, Transactions of the Society of Motion Picture Engineers, vol. 11, no. 23, pp 707-727.
Sheppard S and Houck R (1930) 'The Structure of Gelatin Sols and Gels I. The Viscosity of Gelatin Solutions’, Journal of Physical Chemistry, vol. 34, pp 273-298.
Wilson D (2001) Investigation of Hypersensitised Photographic Films for Astrophotography, Keele University.
The National Film and Sound Archive of Australia acknowledges Australia’s Aboriginal and Torres Strait Islander peoples as the Traditional Custodians of the land on which we work and live and gives respect to their Elders both past and present.


