

Base polymers
Base polymers
The film base is the carrier component of a photographic object and as such must be transparent, tough and flexible.
Over the years many materials have been used for motion picture film base, however only two main types of polymer have been successful — cellulose esters (cellulose acetates and nitrates) and polyester.
Film base polymers
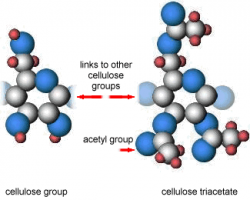
Since the introduction of motion-picture film in the 1890s, the most commonly used base polymers have been esters of cellulose, however polyester is becoming more widely used. Most projection material is now on polyester and it is being more widely used for laboratory stocks.
Cellulose Nitrate
This was the original film base material that was used from 1895 to approximately 1950. Cellulose nitrate was manufactured using cotton linters, a by-product of the cotton industry, that were reacted with a mixture of nitric and sulphuric acids. The resultant polymer was dissolved in solvents, chemicals – such as camphor – added to improve the polymers properties and then 'cast’ on a highly polished flat bed. This produced a thin layer of polymer in solution. The residual solvents evaporated leaving a thin flexible plastic film.
The structure of cellulose nitrate is very similar to cellulose triacetate (Fig 3.1), the main difference is the acetyl groups are replaced by nitro (NO2) groups.
Cellulose nitrate used for film is highly nitrated, this means that the polymer is highly combustible. A common plasticiser used in nitrate film, camphor, is also highly flammable further compounding the problem. This was one of the main drawbacks with nitrate motion picture film. Nitrate film is classed as a 'Dangerous Good’ and as such a license is required to store the film and there are restrictions on transportation.
Nitrate film is soluble in a wide range of solvents. Among these are ether, ethyl and methyl alcohol. One test for nitrate film is its solubility in methyl alcohol – nitrate film base is soluble, acetate and polyester film base is not. Another test compares the specific gravity of nitrate versus acetate base but this uses a solvent that is quite hazardous. The flame test can be used but is not always conclusive as there were additives some manufactures used at times to reduce the flammability of nitrate.
Spectrophotometric testing has shown that nitrate film generally absorbs more towards the blue end of the spectrum than acetate, however this might be indicative of a degree of discolouration (yellowing) due to decomposition.
Positive identification of cellulose nitrate motion picture film therefore needs to be a combination of techniques. Film content that indicates a date of production, edge markings (not print through) and small analytical tests, such as solubility, will provide evidence tending towards nitrate or not.
Cellulose Acetates
These are produced from a cellulose starting material, the most common being wood fibres. The fibres are reacted with a mixture of acetic compounds and sulfuric acid to connect the acetyl groups to the cellulose skeleton.
Additives and solvents are mixed into the raw polymer and the resulting material is cast or extruded to form the thin flexible film.
As film bases, cellulose acetates were first introduced commercially as cellulose diacetate in the early 1910s for 28mm film. Further development of polymers was spurred on by World War I and manufacturing methods became more efficient. The next important development for cellulose acetate film base was the introduction of the 'safety film’ 16mm format for domestic use.
This 'diacetate’ base was still not sufficiently durable for professional use. World War II further improved cellulose acetate polymers and a form, known as 'tri-acetate’, was able to be manufactured with qualities of durability that were equal to the existing professional nitrate film base. Cellulose triacetate was introduced commercially in the late 1940s as solvents needed for its manufacture became more readily available after wartime shortages.
Acetate film falls into two broad categories, 'di’-acetate and 'tri’-acetate. As the names suggest there are varying amounts of acetyl groups in each type. To obtain the desirable properties of strength and durability the cellulose chain needs to be fully esterified or the 'tri’ form. Early manufacturing methods were unable to remove all the residual manufacturing chemicals from the polymer and some of the residual sulfuric acid would be trapped in the structure. This acid would rapidly decompose the polymer. To produce a commercially viable plastic the acid needed to be removed. The process used to do this was to partially hydrolyse the polymer to reduce the number of acetyl groups. This resulted in a plastic with poorer physical characteristics but greater chemical stability. As production methods improved the acetyl percentage was able to be increased.
An early problem with the introduction of cellulose triacetate was finding a suitable film cement as cellulose triacetate has a limited range of solvents. Acetone, methylene chloride and 1-4 dioxane are the major constituents of most current acetate film cements.
Even though c.nitrate was a successful film base the high flammability and cautions required to handle it drove the development of other plastics suitable for film bases. Acetate film is considered a safety or nonflammable base. This is due in part to the lower flammability of cellulose acetate but mainly to the addition of fire retarding agents added during manufacture. Triphenyl phosphate is added to cellulose acetate to serve two functions, that of a plasticiser and even more significantly as a fire retardant. Even so acetate film will burn if sufficient heat is provided, but it does not support combustion in the way nitrate film does.
Polyester (Polyethylene Terephthalate)
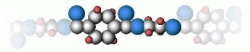
Polyester was developed in the late 1940s. This plastic is created by a condensation reaction between ethylene glycol and terephthalic acid. The ester groups in the polyester chain are polar, with the carbonyl oxygen atom having a slight negative charge and the carbonyl carbon atom having a slight positive charge.
The positive and negative charges of different ester groups are attracted to each other (Fig 3.2). This allows the ester groups of nearby chains to line up with each other forming strong linear structures.
Polyesters do not require plasticisers incorporated in them.

The extreme resistance to tearing makes polyester an ideal film base for the rigours of commercial cinema projection. Even so it is only since the mid 1990s that polyester has become the material of choice for the base of release print materials despite being used for some stills photographic film base since the mid 1950s.
Polyester can be readily identified with a simple non destructive test by examining a clear section of film with two polarising filters, one below and one above the film. By rotating one filter a 'rainbow’ effect can be seen if the material is polyester, other film bases do not show this effect.

Base polymer decomposition
General Cellulose Ester Decomposition
Cellulose ester polymers all suffer from decomposition reactions that will ultimately cause the long molecular chains that form the film base to break apart. The decomposition reactions are similar for both cellulose nitrate and cellulose acetate polymers although the actual by-products and the way these react with the rest of the film may be different. The mechanism of this decomposition is believed to proceed along the following path (cellulose triacetate shown):
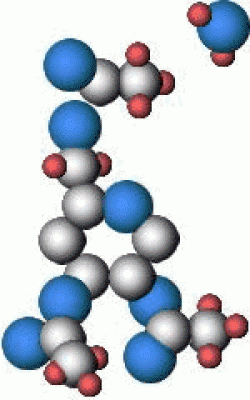
Water. The functional side groups detach in the presence of moisture and heat
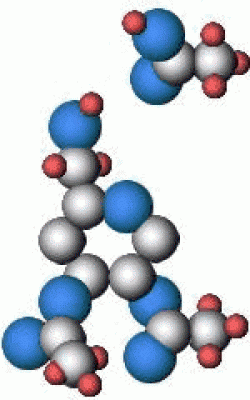
Acetic Acid. These groups then combine with water to form acetic acid
By-products from these reactions are acidic and the acids formed then catalyse, or accelerate, the reaction further. Once initiated this reaction is irreversible.
Cellulose Nitrate
During decomposition of the film base the nitro groups detach themselves from the cellulose base. These groups, as nitrogen oxides, may initially form nitrous acid and then nitric acid by combining with water in the base or emulsion. Nitric acid is a strong acid and consequently the pH can be very low.
The nitric acid produced acts upon the metallic silver that forms the image and eventually converts it into a colourless silver salt, most likely silver nitrate. The acid formed also accelerates the rate of decomposition of the film base. It has been suggested that the acid initiates a digestion reaction of the cellulose in the film base which breaks down the cellulose into sugar-like compounds. This is known colloquially as 'nitrate honey’. The film blocks into a solid mass and finally the base completely disintegrates and turns into a brown powder. Table 3.1 and Fig 3.4 summarise the stages of nitrate decomposition.

Decomposition level: D1
Characteristics: Bleaching of the silver image

Decomposition level: D2
Characteristics: The film becomes sticky

Decomposition level: D3
Characteristics: Bubbles of nitrate 'honey' appear on the surface of the reel

Decomposition level: D4
Characteristics:The film sticks into a solid mass

Decomposition level: D5
Characteristics:The film falls into a brown powder
The bleaching of the image can occur while the film base is still apparently in quite good condition.
Cellulose Acetate (di & tri)
Because the majority of the holdings of most archives are on cellulose acetate based film the major concern of film archives is the decomposition of cellulose acetate known as 'Vinegar Syndrome’. The acid is released within the base but slowly diffuses to the surface causing the noticeable 'vinegar’ odour. Another symptom often noticed in the later stages of 'Vinegar Syndrome’ is the appearance of crystal growths on the surface of the film (Fig 3.5).

These crystals are the plasticisers forced out of the base by a change in the acid content of the base. Analysis has shown that these crystals are mainly triphenyl phosphate (TPP), the most commonly used plasticiser for cellulose triacetate used in film.
In migrating to the surface of the film the plasticisers will form very small holes in the surface of the emulsion.
Occasionally the plasticisers may form small blisters between the emulsion and the base. In extreme cases these will cause the gelatin to rupture and leave a significant hole in the image.
Brittleness and shrinkage are two of the main features of the effect of 'Vinegar Syndrome’. As the level of free acid in the film’s base increases the linkages between the cellulose units can break causing a shortening of the cellulose polymer chains.
This lowers the tensile strength of the base. Shrinkage also occurs as acid migrates to the surface. The base can shrink to a far greater degree than the gelatin emulsion. When this occurs the emulsion layer will shear away from the base and either buckle on the surface or break away from the film in flakes. Early diacetate based films first indicate decomposition by a smell of naphthalene (similar to mothballs). This is the plasticiser, monochloro-naphthalene, being forced from the base by increasing acidity. All other aspects of decomposition of diacetate bases are identical to triacetate although ultimately there are fewer acetyl groups overall to form acetic acid.
Acetic acid is a relatively weak acid and will not cause bleaching of the silver image. However acetic acid is linked to accelerated dye fading in colour films. The shift in pH brought about by decomposition will affect the gelatin. Some degradation of the image structure may occur as the film deteriorates due to softening of the emulsion.
Polyester
Polyester film can decompose with a mechanism very similar to cellulose esters. However this reaction under room temperatures is so slow that it can be considered non-existant. To accelerate this reaction heat or ultra violet light is required.
However there are two potential problems that can occur with polyester base film. Firstly 'core set’ or memory. If film is kept coiled for extended periods of time the base polymer cold flows to reduce the tension and will acquire a high degree of curl. This effect may be minimised by reversing the films wind.
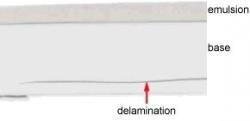
Secondly if the film becomes damp and the emulsion blocks together then the strength of the bond between emulsion layers can be sufficient to cause the polyester to delaminate when it is unwound. Polyester is very strong longitudinally but quite weakly bound as layers (Fig 3.6).

With sufficient force it is possible to cause the film to split and separate. Even if the film does not come apart completely the area that has delaminated will appear as a serious blemish on the image (Fig 3.7).
The National Film and Sound Archive of Australia acknowledges Australia’s Aboriginal and Torres Strait Islander peoples as the Traditional Custodians of the land on which we work and live and gives respect to their Elders both past and present.


