
Shrinkage
The result of moisture and solvent loss in a film.
The film base is manufactured by making a solution of the base material in a solvent. This is then cast onto a moving belt of stainless steel, poured through a slit. The solvent then evaporates, leaving a sheet of base which can be stripped from the belt.
Not all the solvent evaporates in the manufacturing stage — more evaporates subsequently and the film shrinks as a result. This applies to nitrate base and acetate base, but acetate base does not shrink after manufacture to the same extent as nitrate.
Temperature and humidity are the key factors when it comes to shrinkage. If a film is stored in a warm dry atmosphere, it will lose more solvent in a given time than if it is in a cooler, moister atmosphere. It would appear that the practical range of temperature for storage would be between 5°C and 13°C, with 55 per cent relative humidity (RH). At either extreme various problems arise, such as: too high RH permits action of fungus (mould); too low RH increases shrinkage and brittleness. Rather than trying to maintain an exact value that is irregular and with great differences, aim for a constant temperature and humidity.
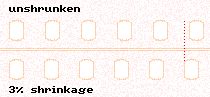
The extent to which a particular film has shrunk can be measured with the use of an instrument called a shrinkage gauge. This consists of a bar on which the film lies. A plate is then laid on top of the film to retain it on the bar and keep it flat. At one end is a fixed pin onto which a perforation is placed. At the other end is a movable pin attached to a dial gauge. The movable pin is moved to the position where the 56th perforation can be placed into it. The dial gauge shows how much the pin needed to move in order to accommodate the shrinkage of the film. We use a ‘setting bar’ to ‘zero’ the gauge. The gauge is marked to show percentage of shrinkage directly.
It is possible to judge the approximate shrinkage by comparing the old with a length (say, 100 perforations) of new film known to be unshrunk.
The National Film and Sound Archive of Australia acknowledges Australia’s Aboriginal and Torres Strait Islander peoples as the Traditional Custodians of the land on which we work and live and gives respect to their Elders both past and present.


